Effects of Acid Rain on Agriculture or Acid Deposition is a broad term that includes precipitation
This has acidic components like sulphuric or nitric acid in it (sulfur dioxide SO2 and nitrogen oxides NOx).
It is harmful to plants, animals, aquatic animals, and even infrastructure.
Acid deposition includes gases i.e. sulfur dioxide and nitrogen dioxide and their derivatives.
As a result of this rain, both dry and wet deposits on soil are affected.
Acid rain affects plants directly and decreases the soil quality to reduce the yield from agriculture.
Moreover, it poses adverse effects in areas that are close to the sources of Sulfur dioxide and nitrogen oxides.
The major cause of acid rain is the presence of sulfur and nitrogen in the atmosphere which mixes with the water vapors and from sulfur dioxide and nitrogen dioxide.
This acid precipitation causes soil erosion affects the yield of crops and vegetation leading to adverse effects.
Emission of Chemicals Leading to Acidification
The presence of sulfur dioxide and nitrogen dioxide leads to the acidification of rain or other forms of precipitation, i.e. acid precipitation.
The normal precipitation has a pH level of 5 and is also slightly acidic.
However, when compound like sulfur and nitrogen mixes in the atmosphere
It makes acid precipitation and falls on the surface of the earth either in the form of rain, sleet, hail, or snow.
If the precipitation becomes too acidic, neutralizing materials in the atmosphere are not able to neutralize the acid component.
Thus leading to acid rain.
Moreover, it results in damage to crops, trees, lakes, rivers, and even animals.
70 Tg(S) comes from burning fossil fuels and industrial smoke each year.
2.8 Tg(S) comes from wildfires and 7-8 Tg(S) from volcanoes.
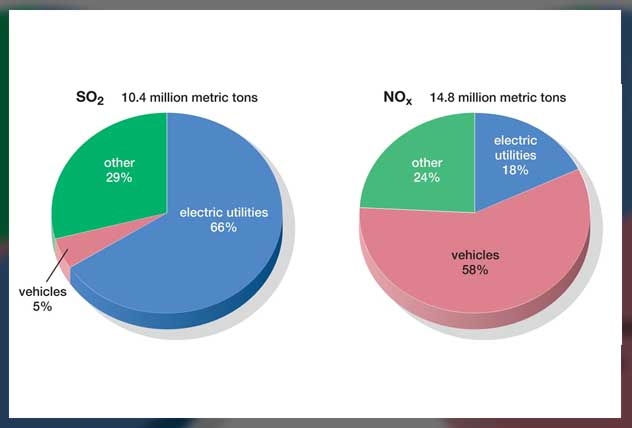
However, it is also important to note that nitrogen from plant life and from an electric activity is the main source of nitric acid in rainwater.
For instance, Fumaroles from the Laguna Callente crater of Poas Volcano create an extremely high amount of acid rain and fog.
As a result of this acid rain, there is an adverse effect on the land and its vegetation and causes irritation to eyes and lung diseases in the settlement.
It is important to note that the cause of acid rain is biological processes that occur in the land, wetlands, and in oceans.
Human Activity
The principal cause of Acid Rain is from human sources like electricity generation, aminal agriculture, factories.
Moreover, motor vehicles emit sulfur and nitrogen compounds that cause acid rain.
The use of coal for power generation is one of the greatest contributors to gaseous and air pollution and is responsible for Acid Rain.

In the past, factories and industries had short funnels which caused problems locally.
Moreover, the gas emitting from the vehicles that we use in our daily routine releases sulfur and nitrogen dioxide into the atmosphere that leading to air pollution.
This air pollution when reacting with moisture or water vapors in the atmosphere forms acid rain.
Moreover, this air pollutions leads to the formation of acidic fog in the atmosphere.
This not only has adverse effects on humans but also on plants and animals.
However, today factories have taller smoke funnels and the dispersal from these causes air pollution to be carried further.
This results in widespread ecological damage.
Formation of Acid Rain
The phenomena of Acid Rain were first studied by a Scottish chemist, Robert Angus Smith
And studied by examining rainwater chemistry near industrial cities in England and Scotland.
Moreover, in the 1950s, many scientists in the USA (United States) started studying this phenomenon.
By the 1970s it became more recognized as a regional environmental issue that was affecting Western Europe and eastern Northern America.
Moreover, not only manmade pollutants are affecting most acidic precipitation, natural disaster like volcanoes are also causing it.
Normal rainwater is slightly acidic.
This is because of the absorption of carbon dioxide from the atmosphere and organic acids from biological activity.

In addition to these, volcanic activity produces sulfuric acid, nitric acid, (sulfur dioxide SO2 and nitrogen oxides NOx).
Hydrochloric acid depends on the emissions from volcanoes.
Other natural resources are the production of nitrogen by lightening and conversion of organic nitrogen by wildfires.
However, the natural sources of acidification cause small changes, and the pH of this precipitation is significantly lower than 5.2.
Other Factors leading to Acid Rain
Burning fossil fuels and smelting of metal ores is one of the biggest causes of Acid rain
And both are human activities. Many power plants use fossil fuels mainly coal to generate electricity.
This use in power plants is contributing to increasing the amount of SO2 and Nox in the atmosphere leading to acid precipitation.
Moreover, along with power plants, vehicles, and oil refiners are also the biggest causes of acid rain today.
It is important to note that burning only fossil fuels accounts for 60% of nitrogen emissions in the atmosphere.
These emissions react with water vapors in the air and form Sulfuric Acid and Nitric Acid. (sulfur dioxide SO2 and nitrogen oxides NOx).
These relations in the atmosphere create wet depositions products.
However, in the gaseous phases, they can also produce Acidic Dry Deposition.
In areas where there is a large consumption of fossil fuels, the acid deposition will occur in areas downwind of emission sources.
The pH in such areas ranges from 4 to 4.5.
In addition to cloud water and fog in such areas, the rain is more acidic and has not only adverse effects on agriculture but also on humans as well.
Effects of Acid Rain
Sulfur dioxide and nitrogen dioxide are not only greenhouse gas that contributes to global warming.
Nitrogen oxide contributes to the formation of ground-level one which is a major pollutant that can be harmful to both plants and animals.
Acidic rain and fog damage the forests especially at higher altitudes as it deprives them of important nutrients.

The effects of acid rain along with environmental stressors, leave trees and plants less healthy, more vulnerable to cold temperatures, insects, and diseases.
These pollutants also inhibit trees’ ability to reproduce as well.
Difference between Wet Deposition and Dry Deposition
Wet deposition occurs when any form of precipitation removes acid from the atmosphere and delivers it to the surface of Earth.
This removes the acid from the clouds and the atmosphere. However, Dry deposition occurs in the absence of precipitation.
This is responsible for as much as 20 – 60% of total acid deposition.
This happens when the particles and gases stick to the ground, plants, and other surfaces and do not react with water vapors.
Effects of Acid Rain on Soil
The biology of soil is adversely affected due to acid rain. Some microbes present in the soil die due to low pH.
The hydrogen ions mobilize the toxins that leach away the minerals and essential nutrients important for crops and agriculture.
Moreover, it leads to Soil Degradation.
Soil Acidification
Soil acidification and acid rain can cause major problems in the soil.
It is important to note that even in minor cases the plants eventually die due to acidic water that lower’s the pH of the plants.

Acidic water enters the plants and causes the minerals to dissolve and get carried away.
This causes the plant to die due to a lack of minerals for nutrition.
In major cases, the same process happens as in minor cases, however at a much faster rate.
Acid rain that falls on the soil and leaves causes drying of the waxy leafy cuticle.
This causes rapid water loss from the plants and the plant eventually dies.
Moreover, the plants that suffer from soil acidification cannot photosynthesize, as it destroys chloroplast organelles in the plants.
Without the process of photosynthesis, a plant cannot create nutrients for its own survival or give out oxygen.
This ultimately affects the species on Earth as there is a depletion of oxygen in the atmosphere and drought.
Effect on Lakes and Streams
The effects of Acid Rain are mostly visible on water bodies that naturally occur in the ecology of our land. i.e. lakes and streams.
This rain deposits in the lakes and streams and when it flows through the soil
This acidic rainwater can leach aluminum from soil clay particles and then flow into lakes and streams.
Moreover, as the water flows towards the lakes and streams, it carries with it other important nutrients thereby affecting the fish in the water bodies.
Although some fish and animals can tolerate slightly acidic water, however, if the pH of water increases by 5.0 the species around the lakes.
Moreover, streams become acid sensitive and are not able to survive.
Effect on Soil
A study suggests that young rootlets and leaf shoots are typically very sensitive to low pH conditions but other aspects of the plant can be harmed as well.
Acid Rain affects the composition and makeup of soil water, which is a main source of nutrients for the plant and soil substrates themselves.
Excess acid in soils can dissolve soil substrate, leading to erosion.
It can cause de-adsorption and migration out of the growth zone of important trace metals and nutrients (especially ammonium ions)
And release to a solution of toxic quantities of other metals (such as Fe).
The effects depend upon the normal mineralogic and organic makeup of the particular soil
As well as other factors such as rainfall amounts and slope of the growing surface.
Effects on Trees
Acid rain causes the trees to die just like other plants.
It leaches out aluminum from the soil.
This is harmful as it removes other minerals and nutrients from the soil that trees need to grow.
Acidic fog and clouds also tend to strip the nutrients from the trees’ foliage at higher elevations leaving them with brown or dead leaves or needles.

As a result, these trees are less able to absorb sunlight, which makes them weak and less able to withstand the freezing temperatures.
Effect on Major Food Crops
A major study suggests that a certain amount of sulfur dioxide on crops shows different degrees of damage to the crops can be divided into
- sensitive crops,
- medium sensitive crops,
- and resilient crops.
Barley, cotton, soybeans, etc belong to sensitive crops, wheat, beans, peanuts belong to medium sensitive crops, white rice, corn, and potato are resilient crops.
Impact of Acid Rain on Crop Preventive Measures
Acid rain pollution is one of the important factors that affect the global economy and social development.
Due to which humans must control acid rain and global acidification, accelerate the process of sustainable development.
The data indicate that acid rain in most parts of the earth is increasing due to the increase of the acidity in the atmosphere.
Now many countries are trying to control acid rain and reduce acid rain damage to human health and the national economy.
Preventive Measures
Increasing Lake Buffer Capacity
Low Sulfur Coal
Energy Alternative
Conclusion
One of the major reasons for the formation of Acid rain is the increase in the amount of sulfuric and nitric acid. (sulfur dioxide SO2 and nitrogen oxides NOx).
It combines with water vapor and comes down as either wet deposition or dry deposition.
Acid Rain has not only adverse effects on the soil but it also has adverse effects on Plant and their growth.
It depletes the soil of its important nutrients and as a result, there are not enough nutrients for plants and agriculture. It is particularly visible in high-altitude areas.
We need to lessen the amount of combustion of fossil fuels and motor vehicles as they contribute to increasing of sulfuric and nitric acid in the atmosphere.






Pingback: Land Pollution: Causes, Effects and Solutions - Al Ardh Alkhadra - Home